Manufacturing, Ripening, Process Control and Grading
15 Process Control
This chapter is as a summary of important process control principles. Table 11.1 illustrates some of these principles by comparing time versus pH profiles for several cheese varieties. Note: in particular the comparison of low to high moisture Cheddar.
Table 15.1. pH versus time profiles for several cheese varieties (Hill, 2005, Emmons and Tuckey, 1967, Reinbold, 1972)
| Operations | Swiss Type | Gouda | Cheddar MNFS 53% | Cheddar MNFS 57% | Feta | Short Set Cottage | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time | pH | Time | pH | Time | pH | Time | pH | Time | pH | Time | pH | |
| Add starter | 0 | 6.60 | 0 | 6.60 | 0 | 6.60 | 0 | 6.60 | 0 | 6.60 | 0 | 6.60 |
| Add rennet | 15 | 6.60 | 60 | 6.55 | 60 | 6.55 | 30 | 6.55 | 60 | 6.50 | 120b | 6.45 |
| Cut | 45 | 6.55 | 90 | 6.45 | 90 | 6.50 | 75 | 6.50 | 120 | 6.30 | c5 – 6 h | 4.80 |
| Drain or dip into forms | 150 | 6.35 | 180a | 6.35 | 210 | 6.10 | 180 | 6.3 | 140 | 6.25 | c 7- 9 h | 5.2 |
| Milling | NA | NA | NA | NA | 360 | 5.40 | 315 | 5.50 | NA | NA | NA | NA |
| Pressing | 165 | 6.3 | 210 | 420 | 5.35 | 390 | 5.45 | NA | NA | NA | NA | |
| Demolding | 16 h | 5.30 | 8 h | 5.40 | 24 h | 5.20 | 10 h | 5.30 | 24 h | 4.60 | NA | NA |
| Minimum pH | 1 wk | 5.20 | 1 wk | 5.20 | 1 wk | 5.10 | 1 wk | 5.20 | 1 wk | 4.50 | NA | NA |
| Retail | 6 mo | 5.6 | 6 mo | 5.6 | 24 mo | 5.50 | 4 mo | 5.30 | 6 wk | 4.55 | 7 d | 5.2 |
a. Draining for Gouda means the first drainage which is followed by washing before moving the washed curd and whey into forms or a press table. b. A small amount of rennet is added to cottage cheese but the primary coagulant is lactic acid produced by the culture. c. The pH at cutting for cottage cheese is around 4.8. Cutting is followed by cooking and multiple washings (1, 2 or 3).
1. The Objectives of Cheese Manufacturing
To maximize returns, the cheese maker must obtain the maximum yields that are consistent with good cheese quality. For example, water and salt are cheaper than milk fat and protein, but you can only have so much cheese moisture and salt (for more on cheese yield see, chapters 18 – 20). With respect to consistent production of high quality cheese, the objectives of the cheese maker are to:
- Develop the basic structure of the cheese.
- Obtain cheese composition required for optimum microbial and enzyme activity during curing. Optimum composition mainly means optimum levels of moisture, fat, pH (lactic acid), minerals, and salt.
For example, the characteristic texture of Swiss cheese is largely determined at the time when the curd and whey are transferred to the press table. At this time, the basic structure (i.e., the manner in which the casein micelles and fat globules are arranged) and chemical composition (especially mineral content) are already determined. You cannot take Swiss curd at this stage and make Cheddar cheese. On the other hand, it is possible to produce both Feta and a Brie type cheese from the same curd.
2. Moisture Control
- Cheese making is a process of removing moisture from a rennet coagulum or an acid coagulum consisting of fat globules (unless the milk is skimmed) and water droplets (whey) trapped in a matrix of casein micelles.
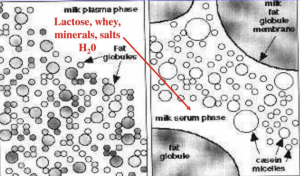
- Cheese is, therefore, a concentrate of milk protein and fat.
- Most cheese making operations are related to this process of removing water from the milk gel by the process of syneresis.
- This liquid (whey) contains water, sugar, whey proteins, lactic acid, and some of the milk minerals.
- The final moisture content, therefore, influences the final pH of the cheese because it determines the residual amount of fermentable lactose in the cheese.
- At the same time, other factors such as the amount and rate of acid development and the temperature and time of cooking, determine the amount and the rate of syneresis.
3. pH Control
- With respect to cheese quality and safety, the most important process control factor is the development of acidity.
- Increasing acidity causes:
- Syneresis (due to reduced charge repulsion on casein micelles) and moisture expulsion.
- Solubilization of calcium phosphates from the micelles.
- Disruption of casein micelle structure with alterations in curd texture.
- Reduced lactose content by fermentation to lactic acid.
- Acid development occurs mainly within the curd because most bacteria are trapped in the gel matrix during coagulation.
- The minimum pH value is dependent on the amount of acid developed during manufacture and the residual lactose that will ferment during early curing and cause further acid development.
- The residual lactose content is mainly determined by the moisture content, washing which removes lactose by leaching, and the extent of fermentation.
- Ability of the culture to ferment galactose is also important. Cultures which metabolize galactose slowly or not at all will be able to produce less lactic acid. For examples, galactose positive strains of Streptococcus thermophilus are selected when limited lactic acid is desired. Similarly Lactobacillus helveticus is used to reduce browning of pizza cheese because it depletes galactose and reduces Maillard browning on the pizza.
- Both the rate of acid development and the amount of acid development (as measured by final pH) are important. For example, the final pH of Swiss is the same as Cheddar, but Cheddar cheese reaches pH 5.2 after about 5 hours while Swiss cheese requires about 15 hours to reach this pH.
- It is important to maintain uniform rate of acid development. If acidity develops too slow or too fast, adjust the amount of culture rather than changing cooking time or temperature.
- pH at draining largely determines the mineral and residual sugar contents of the cheese and from the sugar, the final pH.
- Salting reduces the rate of acid development and therefore, the time and amount of salting is important to prevent the pH from dipping below 5.0 during the first several days after manufacture.
4. Mineral Control
- Loss of calcium phosphate determines extent of casein micelle disruption – hence it determines basic cheese structure. The important parameter is the ratio of Ca to casein or Ca to NFS (non-fat solids), which is easier to measure Table 1.1 column 6 lists Ca in mM/Kg SNF.
- In Swiss (high Ca, about 750 mM Ca/Kg NFS) the casein micelle structure is intact while extensive dissociation and disruption of casein micelles is evident in Feta types (low Ca, about 400 mM Ca/Kg NFS).
- Retention of calcium phosphate also increases the buffer capacity of the cheese, protecting it from big changes in pH.
- pH at draining determines the solubility of calcium and phosphate when the curd is separated from the whey.
- More Ca is retained at high draining pH as in Swiss cheese (pH 6.4 – 6.5) versus Cheddar at pH 6.1 – 6.3 (see table 16.1).
- Calcium retention in soft ripened cheese is low relative to most predominantly rennet coagulated cheese. This is partly due to higher amounts of culture and extended milk ripening and/or setting times, leading to more acid development before cutting. This releases more calcium into the whey. Further, most syneresis occurs during continued acidification overnight in the forms to a final pH of 4.4 – 4.8. The acidic whey caries dissolved calcium salts with it.
When cheese graders refer to cheese texture they often mean the amount and type of openness or holes in the cheese. Here, texture refers to the sensory properties of firmness, elasticity, brittleness, etc.
- Atypical texture in a young cheese is a strong indication of probable flavour defects later. Therefore, a primary objective of cheese making is to develop the ultra-structure that determines the proper texture.
- Conformation of the protein matrix is also influenced by pH. At lower pH, micelles are disrupted, but the proteins are tightly packed because of reduced charge repulsion. Therefore, Feta is brittle while Camembert is soft and smooth due to alkalinity contributed by ammonia during ripening.
- Cheese drained at higher pH retains more Ca and is firmer and more elastic. An example is Swiss types which are inoculated with a small amount of culture to ensure slow acidification in the initial stages. Draining pH is higher (6.3 – 6.4) then most varieties. This helps retain Ca which helps form a curd that is well fused, stretchable and able to form eyes with production of CO2 by Propionibacterium shermanii.
- Later curd handling treatments such as salting and pressing also modify texture and body, but have minimal effects on the basic structure of the protein matrix.
5. Flavour Control
In the broadest terms, directed flavour development depends on retaining or adding ripening agents and controlling their activity over time. Important principles are:
- Milk heating and clarification treatments determine non-starter bacteria in the milk.
- A great deal of direction is obtained through selection of cultures, coagulants and other additives. Debittering cultures for example are an important tool to reduce bitterness and extend the shelf life of high moisture varieties such as Monterrey Jack and of low fat varieties.
- All cooking and curd handling procedures have specific effects on the types of ripening agents (bacteria and enzymes) that remain to ripen the cheese.
- Again, pH at draining is important because it determines the distribution of plasmin and rennin between the curd and the whey.
- Plasmin, an endogenous milk protease, prefers neutral to slightly alkaline pH and is more soluble at low pH. Therefore, varieties that are dipped at high pH, such as Swiss types have higher retention and activity of plasmin. Plasmin activity is also increased by higher cooking temperatures in traditional Swiss and Italian varieties.
- Calf rennet is more soluble at higher pH, but more active at lower pH. Therefore, rennet retention is higher for varieties that are drained at lower pH and in particular for varieties that are cut at lower pH. Rennet activity is also drastically reduced by the higher cooking temperatures in traditional Swiss and Italian types, so it is more active in mesophilic varieties.
Syneresis, which is the contraction of curd to expel water, is controlled mainly by the temperature and time of cooking, as well as the temperature of the wash water. Syneresis increases with higher temperatures during cooking or washing.
principle, natural proteolitic enzyme in milk
Another term for chymosin, the proteolytic enzyme which causes milk coagulation

